Using VSEPR theory draw the structure of XeOF2 and ICl4 -
1) The central atom in XeOF2 is Xe. It carries 8 electrons.Out of which 3 bond pairs and 2 lone pairs (toatal 5 pairs of electron) and hybridization will Sp3d and basic structure is triagonal bipyramidal.But lone pair does not participate in geometry therefore they are missing from 2 vertex of triagonal bipyramidal give it a T-shape as shown in figure.

2) In ICl4− The central iodine contains 8 electron(7 from Iodine and one negative charge) out of which 4 bond pair and 2 lone pair (total 6 pairs of electron) hybridization sp3d2 and basic structure is octahedron.But the lone pair is missed from two opposite vertex of octahedron give it a square planar structure.
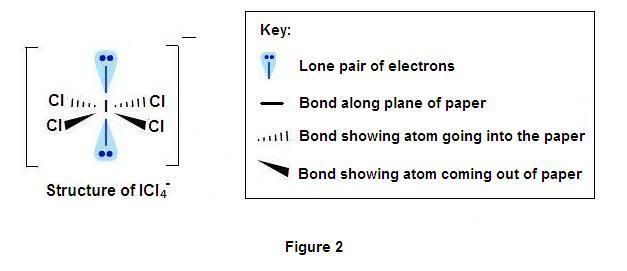

2) In ICl4− The central iodine contains 8 electron(7 from Iodine and one negative charge) out of which 4 bond pair and 2 lone pair (total 6 pairs of electron) hybridization sp3d2 and basic structure is octahedron.But the lone pair is missed from two opposite vertex of octahedron give it a square planar structure.


